Blood fluke infection does not significantly affect the productivity of the SBT industry; however, it has been identified as a concern in ranched Southern Bluefin Tuna (SBT). Two species of blood fluke have been observed to infect SBT: Cardicola forsteri and Cardicola orientalis.
Blood flukes infect Bluefin tuna in the wild; however, it is a relatively rare occurrence. The prevalence of blood fluke immediately after being towed from the wild to the farms ranges from 0 to 10%. Prevalence is defined by the number of fish infected divided by the number of total fish. Approximately 6-8 weeks after transfer into the tuna ranches blood fluke infection can reach a maximum of 100% prevalence. This pattern has been observed since 2004.
LIFECYCLE OF THE BLOOD FLUKE
How the prevalence of the blood fluke can increase so dramatically can be explained by the blood fluke lifecycle. Blood flukes have a two-host lifecycle that requires a definite host (or final host) and an intermediate host. In this case the definite host is the SBT. Recently the intermediate host for C. forsteri has been identified as the terebellid polychaete Longicarpus modestus, and for C. orientalisis it has been postulated to be N. gracilibranchis.
When the fluke enters the intermediate host it is called a miracidium. In the intermediate host asexual reproduction occurs; therefore, from one blood fluke hundreds to thousands of blood fluke can be produced. After asexual reproduction the blood fluke is called the cerica, this is the infective stage of the blood fluke lifecycle. The cercia leaving the intermediate host only have short tails; therefore, they rely on ocean currents to take them to their definite host, the SBT. Once the cercia come into contact with the SBT they penetrate the tuna’s skin and enter its circulatory system. The juvenile blood fluke migrates to the heart where it matures into an adult blood fluke. Adult blood fluke in the heart lay eggs which are subsequently pushed into the circulatory system. The eggs travel via the circulatory system to the gills where they mature into miracidium. Miracidium exit the SBT and infect the intermediate host, starting the cycle again.
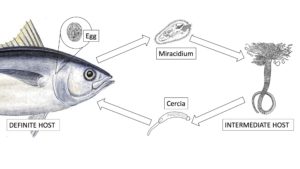
 In tuna ranching, a small number of tuna infected with blood fluke enter the tuna farms where the intermediate host is present. The miracidium from the infected tuna enter the intermediate host where they replicate to high numbers. From the intermediate host a high number of infective blood fluke (cercia) emerge and infect a high number of tuna. This cycle repeats resulting in up to 100% prevalence of blood fluke in tuna.
In tuna ranching, a small number of tuna infected with blood fluke enter the tuna farms where the intermediate host is present. The miracidium from the infected tuna enter the intermediate host where they replicate to high numbers. From the intermediate host a high number of infective blood fluke (cercia) emerge and infect a high number of tuna. This cycle repeats resulting in up to 100% prevalence of blood fluke in tuna.
IMMUNE RESPONSE AGAINST BLOOD FLUKE
SBT that are caught from the wild are generally around 3 years old; therefore, the tuna captured for farming have already survived three years or more of challenges. Consequently, the fish brought to the tuna farms are healthy.
Blood fluke infection is a natural occurrence in the wild; therefore, SBT have evolved an immune response against this parasite. Studies have demonstrated that ranched SBT are capable of mounting a specific humoral response against C. forsteri (Aiken et al., 2008). The antibody response after transfer to tuna farms is delayed which enables the peak in blood fluke burden 6-8 weeks post-transfer. However, as the antibody response increases, the fluke burden decreases.

In the gills of infected SBT, eggs that were killed by the immune response can be seen to be penetrated by phagocytic cells. Occasionally, in infected gill filaments and myocardium (heart muscle) granulomas can be observed surrounding fluke eggs (Colquitt et al., 2001). A granuloma is a structure formed during an immune response against a foreign substance, it essentially “walls off” the infective agent.
Such observations demonstrate the ability of SBT to fight off the infection.
PATHOLOGICAL FINDINGS IN SBT INFECTED WITH BLOOD FLUKE
In the process of the adult blood fluke releasing their eggs into the bloodstream, some eggs become trapped in the myocardium. Other eggs that travel to the gills get stuck and accumulate in the filaments.
Accumulation of eggs in the gills causes significant gill pathology and can obstruct normal blood flow; thus, affecting respiratory efficiency of the fish. In severe cases fish may suffocate to death.
Granulomas form in the myocardium as part of the immune response against trapped blood fluke eggs. This can result in fibrotic tissue. In addition, upon tuna ranching the heart has been observed to enlarge. In severe cases, the accumulation of fibrotic tissue and enlargement of the heart could affect the heart’s ability to pump effectively. The heart rate of the fish may then increase to compensate for the ineffective pumping of the heart. Any extra demand placed on the already weakened heart could cause stress to the fish, especially if ambient oxygen concentrations are low. In severe cases this stress could be sufficient to cause mortality from heart failure.
It is important to note that only in severe cases does death occur.
PRAZIQUENTAL
Blood fluke is a main concern for the SBT aquaculture industry; however, the industry hasn’t been substantially affected by its presence. Nonetheless, minimising the burden from blood fluke is a main priority. In 2011 a treatment for blood fluke was introduced. In 2010 the mortality rate of ranched SBT was around 12.5% (this wasn’t solely due to blood fluke, there are other factors contributing to mortality of fish including stress and predation). Since introducing the treatment we have significantly decreased the mortality rate from 12.5% to 2% in 2012.
The treatment being used is called Praziquantel. Praziquental is an anthelmintic and it works by breaking down the cell walls of the blood fluke, effectively killing it. We inject the tuna’s feed (sardines/pilchards) with praziquental and feed this to the tuna. The treatment gets into the tuna’s bloodstream where it can then go to work on the blood fluke. In using this treatment, we have not eliminated the blood fluke from the tuna farms; however, we are disabling its ability to infect tuna and cause mortalities.
OFFSHORE FARMING
In response to blood fluke infection, the industry has adapted its husbandry techniques to minimise disease burden. For example, farming in deeper water helps reduce blood fluke infection, as the intermediate stages of the blood fluke lifecycle (miracidium and cercia) struggle to move between hosts due to the deeper water column.
There is ongoing research regarding blood fluke infection and how to minimise its burden on tuna health and the industry.